Want cleaner air and safer spaces? Ozone and hydroxyl radicals are two advanced methods for reducing pathogens like bacteria, viruses, and mold in homes, businesses, and vehicles. Here’s what you need to know:
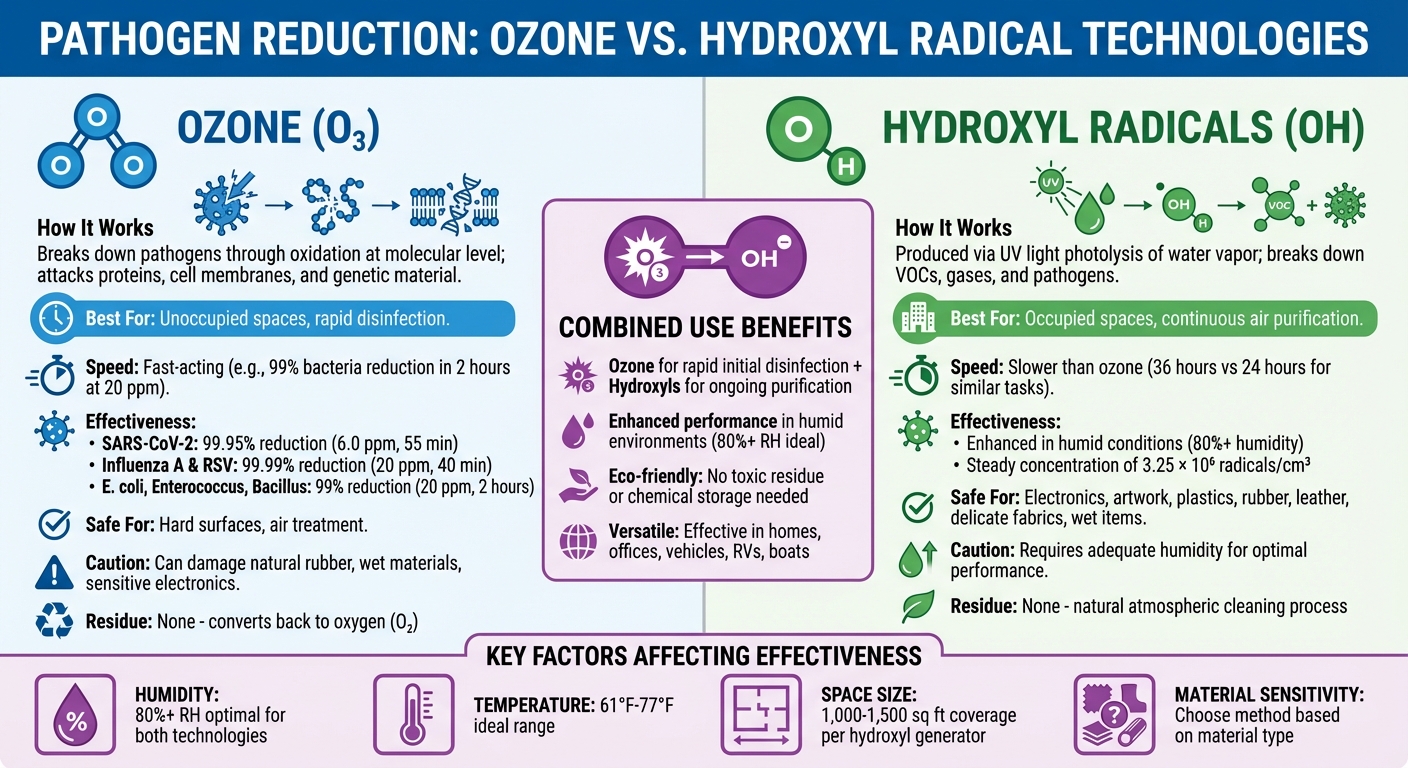
- Ozone: A powerful gas that destroys pathogens by breaking apart their cellular structure. It works quickly but is best for unoccupied spaces.
- Hydroxyl Radicals: Mimic natural atmospheric cleaning processes, breaking down contaminants safely in occupied areas.
- Combined Use: Ozone disinfects surfaces and air rapidly, while hydroxyls provide ongoing air purification, especially in humid conditions.
- Key Benefits: Both methods leave no harmful residue, work on a variety of materials, and can handle hard-to-reach spaces like carpets and air ducts.
These technologies are effective against bacteria, viruses (like SARS-CoV-2), mold, and odors. However, their success depends on factors like humidity, temperature, and the type of space or material being treated. Ozone works faster but can damage sensitive materials, while hydroxyls are safe for delicate items like electronics and artwork.
Quick Tip: Use ozone for deep cleaning in empty spaces and hydroxyls for continuous air quality in occupied areas. Together, they offer a flexible solution for cleaner, healthier environments.
Ozone vs Hydroxyl Radicals: Pathogen Reduction Methods Comparison
The Pros and Cons of UV and Ozone Systems with Chemistry Expert Bob Lowry
sbb-itb-5fea4da
How Ozone and Hydroxyl Radicals Work
Ozone and hydroxyl radicals neutralize pathogens through oxidation, each using distinct mechanisms. When combined, these technologies offer a powerful approach to disinfection. Let’s dive into how each works and the benefits of using them together.
Ozone’s Role in Pathogen Elimination
Ozone (O₃) fights pathogens by breaking them down at a molecular level. It reacts with amino acids and functional groups in proteins, targeting key structures on pathogens. Luigi Cristiano from the Aesthetic and Medical Biotechnologies Research Unit at Prestige explains:
"Capsid surface proteins, as well as membrane receptors present on enveloped viruses, are the first targets of ozone because it reacts directly with amino acids and functional groups of proteins."
Ozone doesn’t stop there. It attacks cell membranes through lipid oxidation and damages genetic material, creating secondary reactive oxygen species (ROS) that further weaken microbial defenses. Once its job is done, ozone naturally converts back to oxygen, leaving no chemical residue behind.
How Hydroxyl Radicals Function
Hydroxyl radicals (OH) are produced when ultraviolet light at 185 nanometers splits water vapor molecules in a process called photolysis. In a controlled environment, such as a 120 m³ (around 4,200 ft³) chamber, commercial hydroxyl generators maintain a steady concentration of approximately 3.25 × 10⁶ radicals per cubic centimeter.
These radicals are highly effective at breaking down volatile organic compounds (VOCs), harmful gases, and pathogens. Their activity increases in humid conditions because more moisture means more hydroxyl radicals are generated. Unlike ozone, hydroxyl generators can run continuously in occupied spaces, improving indoor air quality through ongoing air purification without requiring people to leave the area.
How Ozone and Hydroxyls Work Together
When combined, ozone and hydroxyl radicals tackle disinfection from multiple angles. Ozone works quickly to disinfect surfaces and neutralize airborne pathogens, while hydroxyl radicals provide long-lasting air purification, particularly in humid environments where their production is enhanced. This combination is especially effective in spaces with varying humidity levels, temperature changes, or a mix of surface types.
Typically, ozone is best suited for rapid disinfection in unoccupied areas, while hydroxyl radicals are ideal for continuous air cleaning in spaces where people are present. Together, they create a comprehensive solution for maintaining cleaner, healthier environments.
Pathogens Neutralized by These Technologies
Ozone and hydroxyl radicals are highly effective at neutralizing a wide range of pathogens. Their success, however, depends on the pathogen’s structure and the specific treatment conditions. Let’s break down how these technologies impact bacteria, viruses, and other pathogens.
Bacteria and Viruses
Both technologies are particularly effective against bacteria and viruses, although the degree of success varies by pathogen type. Enveloped viruses, such as SARS-CoV-2 and Influenza A, are especially susceptible because ozone directly attacks their lipid membranes. For example, SARS-CoV-2 viral loads dropped significantly – from 2.0 × 10⁶ PFU/mL to 1.0 × 10³ PFU/mL – when exposed to 6.0 ppm ozone for 55 minutes. Similarly, Influenza A and RSV saw reductions of 99.99% with 20 ppm ozone in just 40 minutes.
Bacteria also respond quickly to these treatments. In a December 2022 study, a Sani Sport ozone device operating at 20 ppm achieved a 99% reduction of E. coli, Enterococcus faecalis, and Bacillus subtilis on medical equipment like syringes and catheters within 2 hours.
When it comes to non-enveloped viruses, treatment requires higher ozone concentrations and increased humidity levels for effective neutralization. For instance, Murine Norovirus achieved a 2 log10 (99%) reduction after 40 minutes at 0.23 ppm ozone under high humidity conditions. Bacterial spores, however, are even more resistant. In May 2020, Joseph Wood from the U.S. EPA demonstrated that Bacillus anthracis (Ames strain) spores required an intense 9,800 ppm ozone concentration at 85% relative humidity for 6 hours to reach a ≥ 6 log10 reduction (99.9999%) on surfaces like wallboard, carpet, and wood.
Fungi and Mold
Fungal spores, with their tough cell walls, demand longer exposure times compared to bacteria and viruses. Ozone and hydroxyl radicals are effective in eliminating mold spores and fungal growth, both on surfaces and in the air. These treatments work by oxidizing the cell walls, which halts reproduction and prevents further spread. When relative humidity exceeds 80%, the spores swell, allowing the gases to penetrate more effectively. This makes these technologies particularly useful in damp environments where mold tends to thrive. Treatment durations depend on the extent of contamination and the type of surface being treated. Porous materials, like textiles, generally require longer exposure times than hard, non-porous surfaces.
Benefits of Combining Ozone and Hydroxyl Technologies
Pairing ozone and hydroxyl technologies creates a powerful two-stage system for reducing pathogens and eliminating odors. Each method has unique strengths that, when combined, deliver better results than using either one alone.
Ozone works quickly in unoccupied spaces, breaking down odors and pathogens at high concentrations. On the other hand, hydroxyl generators are safe for use in occupied spaces, offering continuous treatment without the need for evacuation. This flexibility allows for a strategic approach: ozone can handle intensive initial treatments, while hydroxyls maintain ongoing air quality. For example, during a Manhattan restoration project, hydroxyl treatment successfully eliminated odors from over $1 billion worth of artwork. Together, these technologies offer a well-rounded solution for a variety of situations.
Enhanced Performance in Humid Environments
Hydroxyl generators rely on UVA light and a catalyst to create radicals from water vapor. This makes them especially effective in damp areas, where mold and mildew are common. Similarly, ozone reacts with water to produce oxidizing hydroxyl groups. When used together, these technologies thrive in humid conditions. Moisture supports continuous hydroxyl production, while ozone delivers a fast initial reduction of pathogens, creating a complementary system.
Eco-Friendly and Residue-Free
One of the standout benefits of these technologies is their ability to break down pollutants into harmless byproducts, leaving no toxic residue behind. Ozone naturally reverts to oxygen (O₂) after reacting with contaminants. Unlike chlorine, which can produce harmful byproducts like trihalomethanes, ozone and hydroxyls are clean alternatives. As Tom McArdle, Vice President of Odorox Hydroxyl Group, puts it:
"Hydroxyls… are often referred to as ‘nature’s broom’ because of their crucial role in helping to keep the Earth’s atmosphere clear of harmful organic and inorganic compounds".
Both technologies are generated on-site from oxygen or ambient air, eliminating the need for transporting, storing, or disposing of chemicals.
Versatility Across Different Spaces
This combined approach is effective in a wide range of spaces, from homes and offices to vehicles, RVs, and boats. Ozone’s ability to diffuse four times faster than liquid sanitizers allows it to penetrate hard-to-reach areas like small crevices, textured surfaces, and upholstery. Meanwhile, hydroxyls are safe for delicate materials such as rubber, plastic, leather, electronics, and even artwork, which could be damaged by high ozone concentrations. This versatility makes it an excellent choice for restoration professionals, who can deodorize while simultaneously handling drying, cleaning, and demolition tasks – avoiding costly delays.
For residential applications in the Chicagoland area, companies like Ozonated Cleaning (https://ozonatedcleaning.com) use this dual approach to treat homes, vehicles, and commercial properties. Home treatments typically range from $600 to $900, depending on the size of the space.
Factors That Affect Treatment Effectiveness
The success of ozone and hydroxyl treatments depends on various environmental and situational factors. Recognizing these variables can help achieve better pathogen reduction and biological odor removal.
Humidity and Temperature Effects
Humidity is a key factor in the effectiveness of both ozone and hydroxyl treatments. High humidity levels, ideally above 80%, promote the formation of reactive radicals, which reduces the amount of ozone required and shortens treatment time. On the flip side, dry conditions and higher temperatures reduce ozone stability, making disinfection take longer. For instance, studies reveal that at 85% relative humidity (RH), pathogen inactivation requires significantly lower ozone concentrations than at 55% RH. To illustrate, achieving a 99% reduction of the dsDNA(T7) virus demands a total ozone dose of 114 min[ppm] at 55% RH.
Hydroxyl generation also depends on ambient moisture. In dry conditions, treatment times can stretch out. For example, one test in a 120 m³ (approximately 4,240 ft³) chamber showed a steady-state hydroxyl concentration of (3.25 ± 0.80) × 10⁶ cm⁻³. Temperature is another important variable – ozone is effective across a temperature range of 61°F to 77°F, but higher temperatures cause it to break down faster into oxygen, reducing its efficacy.
To optimize treatment, aim for a relative humidity of at least 55%, with 80% or higher being ideal. If conditions are less than ideal, adjust the total dose (ppm × minutes) accordingly. These factors also influence how long treatments take and how well they distribute, which ties into the next point on space size and layout.
Space Size and Treatment Duration
The size and layout of the area being treated play a major role in determining treatment success. A typical hydroxyl generator can cover 1,000 to 1,500 square feet with ceilings up to about 20 feet high. Ozone treatments, however, tend to work faster; a task that might take 24 hours with ozone could require around 36 hours using a hydroxyl generator in a small enclosed space.
Larger spaces or areas with a heavy pathogen load will need longer treatment times. It’s crucial to maintain a steady oxidant concentration throughout the entire space. Circulating fans can help prevent low-concentration zones caused by stagnant air. In spaces with complex layouts, equipment may need to be relocated to ensure even distribution. Additionally, the sensitivity of materials in the space can influence the treatment approach.
Material Sensitivity Considerations
Ozone has the potential to damage certain materials, especially natural rubber, which it degrades over time. Russ McCubbin, President of International Ozone Technologies Group, highlights this risk:
"Ozone can bleach wet materials and damage natural rubber".
To avoid damage, ensure that natural rubber and wet materials are either removed or completely dried before treatment.
Hydroxyl radicals, while more reactive than ozone, are much safer for delicate materials like electronics, plastics, fine artwork, and fabrics. Tom McArdle explains this benefit:
"It [hydroxyl technology] allows odors to be eliminated from all contents without the risk of damaging them – rubber, plastic, electronics, artwork, wet items, etc.".
This makes hydroxyl treatments especially useful for restoration projects involving valuable items where removing or replacing them would be challenging or expensive.
| Material Type | Ozone Sensitivity | Hydroxyl Safety |
|---|---|---|
| Natural Rubber | High risk of degradation | Safe for treatment |
| Wet Materials | Risk of bleaching | Safe for treatment |
| Electronics | Moderate risk | Safe for treatment |
| Artwork & Fabrics | Moderate risk | Safe for treatment |
Summary
ozone and hydroxyl technologies offer an environmentally friendly and effective way to reduce pathogens and eliminate odors. Both rely on oxidation but are suited for different purposes: ozone works quickly for disinfecting unoccupied areas, while hydroxyl radicals are safe for use in spaces with people present. Together, they provide a versatile solution for pathogen control in a variety of environments.
These technologies are highly effective against bacteria, viruses, fungi, and mold. Studies show that ozone rapidly neutralizes viruses, while hydroxyls penetrate deeply into porous surfaces, addressing pathogens that conventional cleaning methods often miss. Unlike chemical sanitizers, they leave no harmful residues – ozone naturally converts back to breathable oxygen, and hydroxyls replicate the natural cleaning processes found in the atmosphere.
Their performance depends on factors like humidity, temperature, and the size of the area being treated. Material sensitivity is also important: ozone can harm natural rubber and bleach damp fabrics, while hydroxyls are safe for delicate items such as electronics, artwork, and other sensitive materials.
For the best results, treatments should be adjusted based on whether the space is occupied, environmental conditions, and the sensitivity of materials. When used with the right settings, these technologies provide a clean, residue-free, and eco-conscious method for pathogen control in homes, businesses, and vehicles.
FAQs
What are the differences between ozone and hydroxyl radicals in terms of effectiveness and safety?
Ozone (O₃) and hydroxyl radicals (OH•) are both highly effective for reducing pathogens and eliminating odors, but they operate differently and come with distinct safety concerns.
Ozone is incredibly reactive, quickly oxidizing bacteria, viruses, and odors. This makes it a powerful choice for disinfection and deodorization. However, it comes with a significant drawback: it’s unsafe in occupied spaces. High levels of ozone exposure can lead to respiratory problems, so its use requires vacating the area and following strict safety protocols.
Hydroxyl radicals, in contrast, are naturally occurring and short-lived, breaking down almost immediately after neutralizing contaminants. This makes them safe to use in spaces where people are present. Because they don’t accumulate to harmful levels, hydroxyl radicals are perfect for continuous treatment, offering ongoing odor control and pathogen reduction without the need for interruptions. For environments needing uninterrupted safety and cleanliness, hydroxyl radicals provide a practical and safer alternative.
What affects the effectiveness of ozone and hydroxyl treatments for pathogen reduction?
The effectiveness of ozone and hydroxyl treatments hinges on several important factors. First, correct application is absolutely essential. For instance, ozone treatments require the space to be empty during use since ozone can be harmful if inhaled. Also, the equipment used must be appropriately sized and operated to generate enough ozone or hydroxyl radicals to neutralize pathogens effectively.
Environmental conditions significantly influence the outcome as well. Variables like ventilation, humidity, and the treatment’s ability to reach hidden spots – such as air ducts or tight crevices – can affect how well it works. Proper air circulation ensures these reactive agents can cover all surfaces. Lastly, adhering to safety protocols is non-negotiable. This includes thoroughly ventilating the space after treatment to ensure safety and effectiveness. When these factors are carefully managed, ozone and hydroxyl treatments can deliver excellent results in reducing pathogens and eliminating odors.
Is it safe to use ozone and hydroxyl technology around electronics and delicate materials?
Ozone and hydroxyl technologies can be effective, but their use requires careful attention, especially around electronics and fragile materials. Ozone, for instance, is a powerful oxidizer that can harm items like rubber, electrical wiring, and certain plastics if used in high concentrations or for too long. To avoid damage, ozone generators should only be operated in unoccupied spaces, with proper ventilation and strict adherence to safety protocols.
Hydroxyl technology, by contrast, is often seen as a safer option for use around sensitive materials. Hydroxyl radicals are less reactive than ozone and are less likely to cause material degradation when used as directed. Even so, it’s essential to follow manufacturer instructions and consult with experts when treating areas that contain delicate or electronic items.


Recent Comments